Ozone
Ozone (O3) is a form of oxygen generated by reactions between sunlight and oxygen (O2). This process is most efficient in the stratosphere at altitudes of 15–50 kilometres. Ozone is carried by natural air motions and destroyed by chemical cycles. As a result, its concentration is highly variable in space and time. The thickness of the ozone layer is significant because ozone absorbs ultraviolet (UV) radiation from the sun, acting as a filter against sunburn and other damage. Without the protection of the ozone layer in the stratosphere, terrestrial life as we know it could not survive.
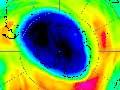
The amount of ozone in an atmospheric column up to 50 or 60 kilometres is measured in Dobson Units (DU), where 1 DU corresponds to a layer of pure ozone 0.01 millimetre thick at surface temperature and pressure. The global average ozone is 300 DU. Over New Zealand, the ozone layer ranges from about 250 DU in autumn to 450 DU in spring.
Ozone depletion
Ozone can be destroyed by reactions with by-products of man-made chemicals, such as chlorine from chlorofluorocarbons (CFCs). Increases in the concentrations of these chemicals have led to the much-publicised ozone depletion.
Effects of ozone depletion
Decreased ozone can have serious environmental impacts by increasing UV radiation at the ground. In humans, this causes eye damage (cataracts) and skin damage (sunburn and skin cancer). It has been calculated that for each 1% reduction in ozone there is an increase in UV radiation of about 1.2%, which could lead to a 2% increase in skin cancers. It also harms plants and animals, and damages materials such as plastics and paints.
Ozone depletion in New Zealand
The most dramatic ozone losses occur in Antarctica, where spring ozone columns can be less than 90 DU. This ‘ozone hole’ lies well to the south of New Zealand and does not pose a direct health risk. However, when it breaks up, filaments of ozone-poor air can sometimes pass overhead.
New Zealand’s summertime ozone has decreased by about 10% since 1970. During the first half of the 21st century it is expected to recover, albeit slowly, because the release of the damaging chemicals is now restricted by the Montreal Protocol on Protection of the Ozone Layer. The rate of recovery may be influenced by the changing climate.
Land of rising sun damage
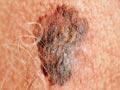
Living in what has been called the melanoma capital of the world, Aucklanders face a 5.7% chance of developing skin cancer. Although the city's climate encourages outdoor activities, in summer UV intensities are high due to the low ozone and clean air, and because the earth is closer to the sun. People with fair skin are particularly susceptible to being harmed by the sun's radiation.
Skin cancer rates
New Zealand’s death rate from skin cancer is about 300 per year, the highest in the world relative to population (and over half that from road accidents). This is due to the relatively high UV exposures and the high number of fair-skinned people. Peak UV intensities in New Zealand are about 40% greater than at comparable latitudes in Europe.
The UV Index (UVI) represents the intensity of UV radiation. A UVI greater than 10 is extreme, and skin damage can occur in less than 15 minutes. In New Zealand, the midday UVI can exceed 13 in summer. In winter, it rarely exceeds 2, although intensities increase with altitude and when the surface is snow-covered, as on skifields.
The most important factor controlling UV radiation is the sun’s angle above the horizon. At low angles, the rays pass through much more of the atmosphere, reducing the radiation. As a rule of thumb, if your shadow length exceeds twice your height (when the angle of the sun is less than 30°), then there is little risk of UV damage. If your shadow is less than your height (the sun angle is greater than 45°), then protective measures are needed.